Device compliance sustained
95%
Across wearable-enabled studies with active support
Unmonitored sync lag
3 days
Typical before failures are caught without an operating layer
Support model
Proactive
Issues identified and resolved before signal loss normalizes
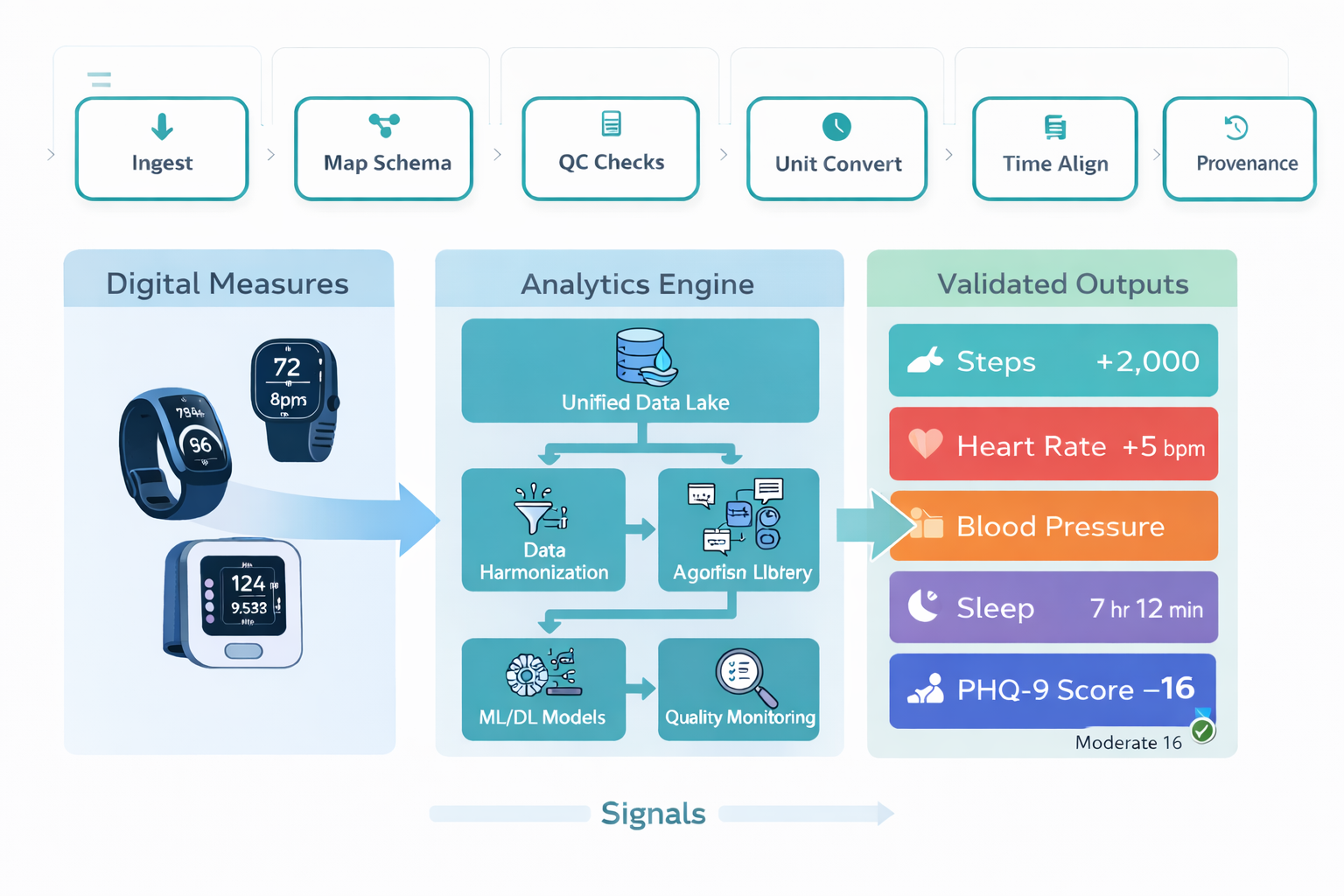
Wearable Compliance Layer
Monitor → detect → resolve → protect
Where Wearable Compliance Breaks Down
Wearable compliance degradation follows predictable patterns. Most programs lose data not because participants refuse to participate, but because small operational failures accumulate without intervention.
- Charging fatigue: participants stop charging consistently after the first few weeks, creating recurring data gaps
- Sync failure: Bluetooth connectivity issues, app permissions, and OS updates silently interrupt data transmission
- Non-wear events: participants remove devices during activities without recording the gap
- Device malfunction: hardware issues that go unreported because participants do not know how to escalate
- App friction: updates, login issues, and confusing interfaces reduce manual data submissions
- Study fatigue: participants who are otherwise engaged begin treating the wearable as optional
Without a monitoring layer that catches these patterns early, individual gaps become accepted loss and accepted loss compounds into compromised endpoint integrity.
Wearable Compliance Requires More Than a Dashboard
Compliance dashboards show where data is missing. They do not fix the reason it is missing, re-engage the participant, replace a broken device, or recover the relationship before the participant decides to stop wearing.
Resolving wearable compliance gaps requires an operating model that connects visibility to action.
- Live compliance monitoring across wear time, sync frequency, and data receipt
- Proactive outreach when patterns signal early disengagement or device issues
- Device troubleshooting and replacement workflows that do not depend on site staff availability
- Participant education that reinforces wear importance across the study, not just at onboarding
- Signal quality review that separates genuine non-wear from artifact before data reaches analysis
Delve Health operates this model so study teams get compliance data that is both complete and trustworthy.
Digital Endpoints Depend on Compliance Quality
As wearable-derived endpoints move into regulatory submissions, data completeness and compliance quality become as important as the endpoint itself. A digital endpoint supported by incomplete wear data is difficult to defend.
- FDA digital health guidance increasingly requires documentation of wear time and data completeness
- Missing data imputation strategies depend on the patterns and magnitude of data gaps
- Endpoint integrity is only as strong as the compliance infrastructure behind the collection
- Post-hoc cleaning of wearable data is costly, time-consuming, and not always recoverable
Investing in compliance infrastructure before data loss occurs is substantially less expensive than trying to recover or justify it during analysis.
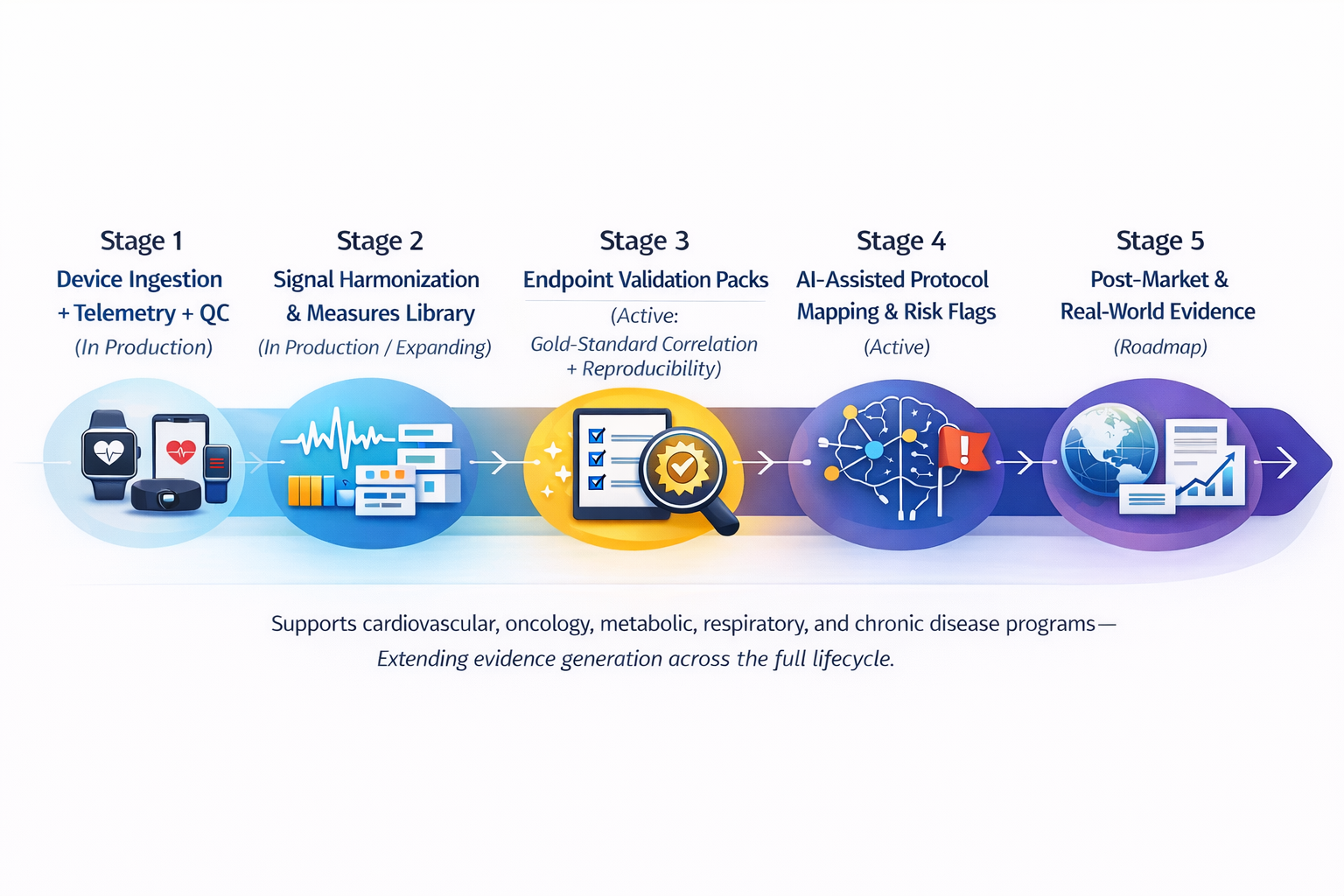
The Four Pillars of Wearable Compliance
Sustained device compliance requires monitoring, support, analytics, and integration working together as a single operating model.
Device Monitoring Across the Study
Track wear time, sync events, and device health in real time to catch compliance degradation before it becomes data loss.
Concierge Device Support
Trained teams handle troubleshooting, re-onboarding, and participant education so device friction does not become dropout risk.
Signal Quality Control
Review raw wearable data for artifacts, anomalies, and non-wear patterns before they contaminate analysis-ready datasets.
Compliance Analytics and Reporting
Study-level and participant-level compliance trends that give sponsors and sites the visibility needed to act before patterns solidify.
Frequently Asked Questions
What is wearable compliance in clinical trials?
Wearable compliance refers to the percentage of expected wear time and data transmissions that are actually captured across a study. High compliance means participants wear devices consistently and data syncs reliably throughout the protocol.
Why does wearable compliance degrade over time?
Charging fatigue, sync failures, device friction, app issues, and general study fatigue all contribute to declining wear time. Without active monitoring and support, degradation goes undetected until significant data is already lost.
Is a wearable dashboard enough to maintain compliance?
No. Dashboards show where gaps exist, but they do not resolve the reasons behind them. Restoring compliance requires outreach, troubleshooting, and a support model that owns device issues from detection through resolution.
How does Delve Health improve wear time in clinical trials?
Delve monitors wear time and sync frequency in real time, triggers concierge outreach when patterns signal emerging compliance issues, handles device troubleshooting, and applies signal quality control to protect endpoint integrity.
What wearable devices does Delve support?
Delve integrates with a broad range of devices including Apple Watch, Fitbit, Garmin, ActiGraph, Biosensors, and others. Device support is matched to protocol requirements through a fit-for-purpose selection process.
Wearable Data Quality Starts With Compliance Infrastructure
If your digital endpoint depends on reliable wear time and data continuity, compliance must be actively owned from the first device shipment.
Review Your Wearable Compliance Model